In 1909, more information about the electron was uncovered by American physicist Robert A. Listen to Thomson describe his discovery in his own voice. 
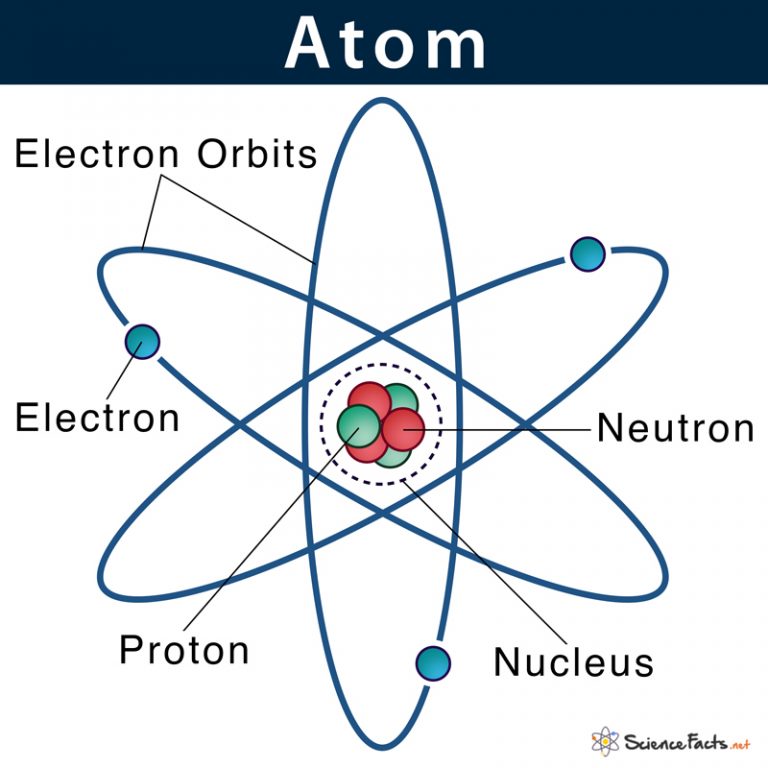
Thomson Talks About the Size of the Electron It was J.J Thomson who took this theory and orchestrated scientific experiments using the cathode rays to prove atoms contained charged particles – the fundamental unit of charge – the electron. The term “electron” was initially coined in 1891 by Irish physicist George Stoney, meaning “electric ion.” Stoney originally recognized the atom must have a unit of electricity and charge associated with the atom, but he had no experimental proof. they are less massive than atoms and indistinguishable, regardless of the source material, so they must be fundamental, subatomic constituents of all atoms.Īlthough controversial at the time, Thomson’s idea was gradually accepted, and his cathode ray particle is what we now call an electron, a negatively charged, subatomic particle with a mass more than one thousand-times less that of an atom.The particles are attracted by positive (+) charges and repelled by negative (−) charges, so the particles of the cathode ray must be negatively charged since like charges repel and unlike charges attract.(credit a: modification of work by Nobel Foundation, PD credit b: modification of work by Eugen Nesper, PD credit c: modification of work by Kurzon, PD).īased on his observations, here is what Thomson proposed and why: Simultaneous deflections by applied electric and magnetic fields permitted Thomson to calculate the mass-to-charge ratio of the particles composing the cathode ray. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Thomson produced a visible beam in a cathode ray tube. Figure 5.3a Thomson Cathode Ray Experiments: (a) J. The results of these measurements indicated that these particles were much lighter than atoms (Figure 5.3a). In similar experiments, the ray was simultaneously deflected by an applied magnetic field, and measurements of the extent of deflection and the magnetic field strength allowed Thomson to calculate the charge-to-mass ratio of the cathode ray particles. This beam was deflected toward the positive charge and away from the negative charge, and was produced in the same way with identical properties when different metals were used for the electrodes. When high voltage was applied across the electrodes, a visible beam called a cathode ray appeared between them. This apparatus consisted of a sealed glass tube from which almost all the air had been removed the tube contained two metal electrodes. In the late 1800s, a number of scientists interested in questions like these investigated the electrical discharges that could be produced in low-pressure gases, with the most significant discovery made in 1897 by English physicist J. The discovery of these subatomic particles is discussed next. If the matter were composed of atoms, what were atoms composed of? Was it just predominately European scientists questioning the properties of matter, its constituents, its behaviour, and why and how it exists? Or at that time, were other groups of people around the world, such as Indigenous communities, also asking similar questions? From their experiences, traditions, and cultural practices did they test their own hypotheses about the different matter around them – what makes mud, mud, and why is it different from sweetgrass before and after it is burned? Did they, too, ask the question what makes the smallest particles, or is there something smaller? The matter is made up of inconceivably small atoms, and yet scientists found atoms contain even smaller subatomic particles, including electrons, protons, and neutrons. Atomic Theory after the Nineteenth Century While the historical persons and dates behind these experiments can be quite interesting, it is most important to understand the concepts resulting from their work. Here, we will discuss some of those key developments, with an emphasis on the application of the scientific method, as well as understanding how the experimental evidence was analyzed. Much of this came from the results of several seminal experiments that revealed the details of the internal structure of atoms. Scientists have made significant progress in furthering our understanding of atomic theory. Describe the three subatomic particles that compose atoms.Summarize and interpret the results of the experiments of Thomson, Millikan, and Rutherford.Outline milestones in the development of modern atomic theory.By the end of this section, you will be able to:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
